INTRODUCTION:
Organ printing is one of the latest advancements in the medicinal field for transplantation using engineering technology. During transplantation, graft rejection is one of the major concerns due to the recipient’s immune response to the donor organ [1]. Thus, the tissue engineering approach for bioprinting helped to overcome this challenge. It can be more precisely specified as layer-by-layer additive manufacturing or Rapid prototyping (RP). Organ printing can be defined as the construction of the organ and biofabrication (assembling biomaterials for tissue engineering) of 3D tissue by using computer aided technology on the basis of digital models [2]. Organ printing is performed according to the patient’s tissue features that explain the chemical composition and complexities of the tissue while handling transplantations. Information technology plays a crucial role in accomplishing bioprinting as for instance, using CAD (Computer Aided Design) software for designing the digital models. In order to execute 3D bioprinting technology, both hardware and the software is required, where 3D bioprinter is the hardware and digital model is the software. A 3D bioprinter is a robotic device that accurately places the tissue spheroids or living cells, rods, and biomaterials according to the digital model in 3-dimensional space. Software is employed for the purpose of organ construction and facilitates 3D bioprinting of tissues. There are three important steps involved in a bioprinting process: pre-processing, processing, and post processing. The pre-processing step includes the tissue and organ imaging along with the organs reconstructed from the image generated. There are several imaging methods used in layer-by-layer additive manufacturing consist of Magnetic Resonance Imaging (MRI), Positron emission tomography (PET), Ultrasonography, Single-photon emission computed tomography (SPECT), Cone beam computed tomography (CBCT) where Multidetector Computed Tomography is commonly used because of its simpler image processing requirements [3]. The processing step uses a suitable bioink for the bioprinting process [4-6]. The bioprinting process is categorized into four different processes: droplet based bioprinting, stereolithography-based bioprinting, laser based bioprinting, and extrusion based bioprinting [7-9]. The post processing process implicates the development of bioprinted tissue [10]. Bioprinting has various applications and has great potential in modern research as well as in the medicinal field.
METHODOLOGY:
As there are 3 major steps involved in the bioprinting process i.e Pre-processing, processing, post processing, following each step are explained in detail.
1] Pre-processing:
Initially, the design outline of the organ to be fabricated has been framed in the form of a computer-aided design. Information regarding the histological structure, composition, anatomy and topology of the organs is required as it is essential to define the spatial configuration and localization of the cells to construct 3D organs. To achieve the anatomical characteristics, clinical bioimaging processes and ultrasound are utilized when the organs are still inside the owner. Thus a patient’s specific anatomical information can be obtained. But by this method, the cellular and histological resolutions can’t be acquired. Hence, a second approach is applied which is based on the reconstruction of the histological section using computer technology. This method produces excellent information about the composition, structure and size of the organ with high resolution. The only disadvantage of the computer aided method is, it can be employed only after the demise of the individual, and human organs tend to change and distort. As organs have polymeric structure i.e have repeating units of the cells, it can be reconstructed by adding the units according to the anatomical structure and assembling the organ in silico. A third approach which can be used is based on the mathematical computational anatomical model by understanding the vascular branching of the organ for creation of realistic models. Once anatomical features of the organ are obtained from bioimaging and attaining histological information from computer-aided design, mathematical modelling can be further used for generating desirable organ blueprint models by gathering micro and macro anatomical information together. In organ printing technology, tissue spheroids are used as building blocks. In the pre-processing stage, biofabrication forms an integral part as tissue spheroids are used to produce organs of desirable size and composition.
Dr. Ken Brakke, the developer of the software “surface evolver” was able to exhibit the change from the cell aggregates to more densely packed tissue spheroids, the diameter of the tissue spheroids was reduced to 20%. So, in this stage mathematical model and computer technology is very important for the simulation of tissue spheroid packaging for final size and shape of the bioprinted organ.

2] Processing:
In the processing step, the approach is the developing virtual organ biofabrication line by applying information technology tools. For obtaining a virtual organ biofabrication line, it is essential to combine various machinery processes and devices, so that real time animation and data can be generated using a computer system providing virtual reality to organ biofabrication plants. This helps to visualize and observe virtually all the components of the organ biofabrication line. This first step can be achieved by using robotic bioprinters, cell sorters, perfusion bioreactors and robotic tissue spheroid biofactricator which are important biofabrication equipment through computer simulation. In the next step, according to the biofabrication process, the equipment is placed and carried out by imposing relevant regulatory requirements. The virtual biofabrication equipment governs the later stages of bioprocessing and organ bioassembly on microlevel. Important tools for development of organ bioprinting are computer simulated, computer aided design, mathematical modelling, information technologies and virtual reality methods.
3] Post-processing:
In the post processing stage, the major task is to ensure survival and viability of the bioprinted organs, fusing of tissue spheroids into integrated constructs and its maturation. This processing will facilitate integration, viability and monitoring of the 3D bioprinted organs. Computing approaches are implemented for simulation of tissue spheroid fusion and other approaches using surface evolver software. Thus large construction of 3D organs is possible by fusion of tissue spheroids. The only limitation of using Surface Evolver software is the difficulty in changing the topological position of tissue spheroids within the 3D construct. In this process another issue arises that is designing inter spheroidal perfusion modelling which requires systematic analysis. The modelling of tissue maturation requires determination of the structural features of the material properties of organs and tissues. The main determinant of the vascular wall is the extracellular matrix which consists of elastin and collagen. One of the studies demonstrated the identified parameters of collagen such as concentration, diameter, level of cross linking that determines the structural and material properties of heart valves. Thus, investigation of this approach will be advantageous.
TYPES OF BIOPRINTING METHODS:
1] LASER BASED BIOPRINTING:
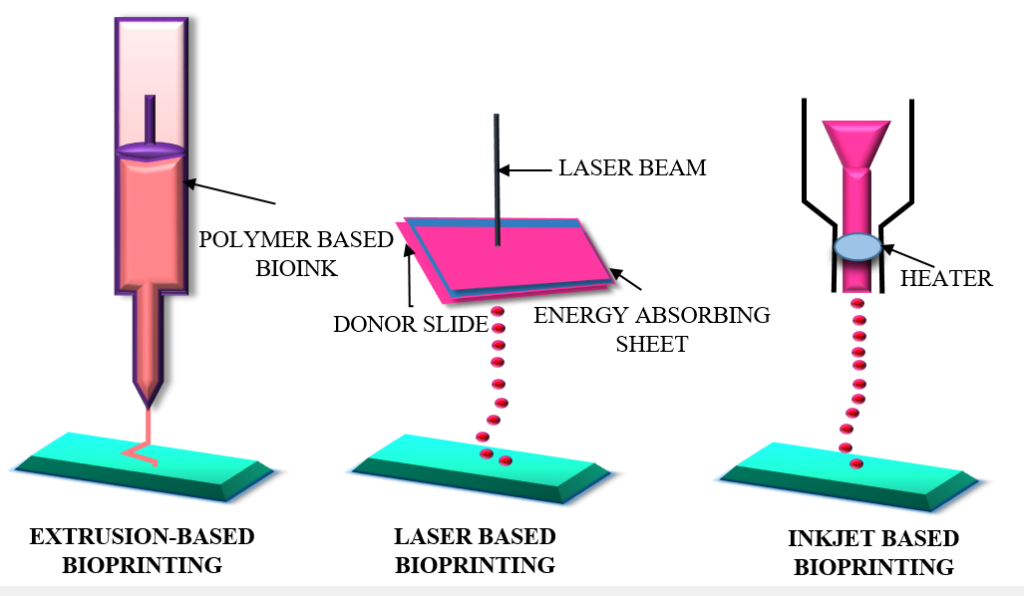
A laser based bioprinter consists of a laser source, a substrate or collector slide and a laser transparent print that is ribbon coated with a layer of cell charged with bioink. The method used in the laser bioprinting approach has high reproducibility and resolution, making it accessible to be used in biomedical fields [11]. With the assistance of computer aided design and manufacturing (CAD/ CAM), the laser energy is focused on patterning the cell laden bioinks into 3 dimensional arrangements. This method is described for printing skin tissue, biopapers, and stem cell grafts [12].
ADVANTAGES:
- High resolution
- High cell viability
- Low viscosity cell suspensions.
- High cell densities
DISADVANTAGES:
- High cost of laser system
- Fabrication of the laser print ribbon
- Laser exposure leading to cell cytotoxicity
- Scalability limitation
- Complications in handling the laser pulses.
2] DROPLET BASED BIOPRINTING:
In this process, the cell laden bioink is released from a nozzle in the form of droplets onto a substrate [13]. For biological as well as non-biological applications, inkjet printers are commonly employed that use thermal forces to eject liquid drops on the substrate. [10].
ADVANTAGES:
- High resolution (20–100 m)
- High printing speed (1–10,000 droplets/s)
- Affordability
- Cell concentration gradient
DISADVANTAGES:
- Nozzle clogging
- Low viscosity bioink
- Droplets cannot be regulated precisely.
3] STEREOLITHOGRAPHY BASED BIOPRINTING:
Light irradiated in stereolithography is usually UV light. In stereolithography based bioprinting based on gelatin methacryloyl (GelMA) with eosin Y (EY) photoinitiation visible-light cross-linkable and cell attachable bioinks were developed for 3D bioprinting and were utilized to print cell laden hydrogels [5]. At high resolutions stereolithography was used for the preparation of 3D hydrogel structures utilizing biodegradable poly (ethylene glycol)/poly (D, L-lactide) materials [14]. Applying this method, 3D printing of cell-encapsulated hydrogels was shown successfully [15].
ADVANTAGES:
- Very high resolution
- High cell concentration
- Reduced printing time
DISADVANTAGES:
- Effect of UV rays on cells damaging the DNA leading to cell lysis.
- Use of only photocurable bioinks
4] EXTRUSION BASED BIOPRINTING:
In extrusion based bioprinting, mechanical force or pneumatic pressure are applied for releasing the bioink from the nozzle.
ADVANTAGES:
- Scalability because of the large deposition rate and continuous flow of bioink.
- High cell concentration
- High viscosity bioinks
- Low cost and simple is execution
DISADVANTAGES:
- Nozzle clogging
- Low resolution
- Shear thinning bioink.
APPLICATIONS OF 3D BIOPRINTING:
- Widely used in tissue engineering.
- Materials such as gelatin, Hyaluronic acid and alginate were used to construct heart valve tissue using extrusion based bio printing technique where the cells were of Aortic valve interstitial cell and Aortic root sinus smooth-muscle cell [16].
- The Jetting based bio printing was used to construct Musculo-skeletal tissue using fibrin and growth factors such as BMP-2, FGF-2 and the cell type in this construction are Muscle-derived stem cells, Myoblast, Mesenchymal fibroblast [17].
- Advancement in medical technologies for the purpose of transplantation of organs or tissues.
REFERENCES:
1] Rustad KC, Sorkin M, Levi B, Longaker MT, Gurtner GC. Strategies for organ level tissue engineering. Organogenesis. 2010 Jul-Sep;6(3):151-7. doi: 10.4161/org.6.3.12139. PMID: 21197216; PMCID: PMC2946046.
2] Mironov V, Kasyanov V, Drake C, Markwald RR. Organ printing: promises and challenges. Regen Med. 2008 Jan;3(1):93-103. doi: 10.2217/17460751.3.1.93. PMID: 18154465.
3] Rengier F, Mehndiratta A, von Tengg-Kobligk H, Zechmann CM, Unterhinninghofen R, Kauczor HU, Giesel FL. 3D printing based on imaging data: review of medical applications. Int J Comput Assist Radiol Surg. 2010 Jul;5(4):335-41. doi: 10.1007/s11548-010-0476-x. Epub 2010 May 15. PMID: 20467825.
4] Gopinathan J, Noh I. Recent trends in bioinks for 3D printing. Biomater Res. 2018 Apr 6;22:11. doi: 10.1186/s40824-018-0122-1. PMID: 29636985; PMCID: PMC5889544.
5] Wang Z, Kumar H, Tian Z, Jin X, Holzman JF, Menard F, Kim K. Visible Light Photoinitiation of Cell-Adhesive Gelatin Methacryloyl Hydrogels for Stereolithography 3D Bioprinting. ACS Appl Mater Interfaces. 2018 Aug 15;10(32):26859-26869. doi: 10.1021/acsami.8b06607. Epub 2018 Aug 1. PMID: 30024722.
6] Gungor-Ozkerim PS , Inci I , Zhang YS , Khademhosseini A , Dokmeci MR . Bioinks for 3D bioprinting: an overview. Biomater Sci. 2018 May 1;6(5):915-946. doi: 10.1039/c7bm00765e. PMID: 29492503; PMCID: PMC6439477.
7] Hong JM, Kim BJ, Shim JH, Kang KS, Kim KJ, Rhie JW, Cha HJ, Cho DW. Enhancement of bone regeneration through facile surface functionalization of solid freeform fabrication-based three-dimensional scaffolds using mussel adhesive proteins. Acta Biomater. 2012 Jul;8(7):2578-86. doi: 10.1016/j.actbio.2012.03.041. Epub 2012 Apr 2. PMID: 22480947.
8] Guillotin B, Souquet A, Catros S, Duocastella M, Pippenger B, Bellance S, Bareille R, Rémy M, Bordenave L, Amédée J, Guillemot F. Laser assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials. 2010 Oct;31(28):7250-6. doi: 10.1016/j.biomaterials.2010.05.055. Epub 2010 Jul 2. PMID: 20580082.
9] Agarwala S, Lee JM, Ng WL, Layani M, Yeong WY, Magdassi S. A novel 3D bioprinted flexible and biocompatible hydrogel bioelectronic platform. Biosens Bioelectron. 2018 Apr 15;102:365-371. doi: 10.1016/j.bios.2017.11.039. Epub 2017 Nov 16. PMID: 29172145.
10] Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014 Aug;32(8):773-85. doi: 10.1038/nbt.2958. PMID: 25093879.
11] Schiele NR, Corr DT, Huang Y, Raof NA, Xie Y, Chrisey DB. Laser-based direct-write techniques for cell printing. Biofabrication. 2010 Sep;2(3):032001. doi: 10.1088/1758-5082/2/3/032001. Epub 2010 Jul 12. PMID: 20814088; PMCID: PMC4887083.
12] Kawecki F, Clafshenkel WP, Auger FA, Bourget JM, Fradette J, Devillard R. Self-assembled human osseous cell sheets as living biopapers for the laser-assisted bioprinting of human endothelial cells. Biofabrication. 2018 Apr 30;10(3):035006. doi: 10.1088/1758-5090/aabd5b. PMID: 29638221.
13] Vijayavenkataraman S, Yan WC, Lu WF, Wang CH, Fuh JYH. 3D bioprinting of tissues and organs for regenerative medicine. Adv Drug Deliv Rev. 2018 Jul;132:296-332. doi: 10.1016/j.addr.2018.07.004. Epub 2018 Jul 7. PMID: 29990578.
14] Elomaa L, Pan CC, Shanjani Y, Malkovskiy A, Seppälä JV, Yang Y. Three-dimensional fabrication of cell-laden biodegradable poly(ethylene glycol-co-depsipeptide) hydrogels by visible light stereolithography. J Mater Chem B. 2015 Nov 14;3(42):8348-8358. doi: 10.1039/c5tb01468a. Epub 2015 Sep 11. PMID: 29057076; PMCID: PMC5650242.
15] Chan V, Zorlutuna P, Jeong JH, Kong H, Bashir R. Three-dimensional photopatterning of hydrogels using stereolithography for long-term cell encapsulation. Lab Chip. 2010 Aug 21;10(16):2062-70. doi: 10.1039/c004285d. Epub 2010 Jul 5. PMID: 20603661.
16] Duan B, Hockaday LA, Kang KH, Butcher JT. 3D bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels. J Biomed Mater Res A. 2013 May;101(5):1255-64. doi: 10.1002/jbm.a.34420. Epub 2012 Sep 27. PMID: 23015540; PMCID: PMC3694360.
17] Phillippi JA, Miller E, Weiss L, Huard J, Waggoner A, Campbell P. Microenvironments engineered by inkjet bioprinting spatially direct adult stem cells toward muscle- and bone-like subpopulations. Stem Cells. 2008 Jan;26(1):127-34. doi: 10.1634/stemcells.2007-0520. Epub 2007 Sep 27. PMID: 17901398.
